
Raman Spectroscopy for Lead Acid Battery Analysis
Lead Acid Battery Analysis Using the Raman Spectrometer
Lead-acid batteries remain a cornerstone of energy storage technology, valued for their reliability, low cost, and high recyclability, particularly in automotive and stationary applications. Despite their maturity, these batteries face performance limitations and degradation mechanisms such as negative plate sulfation, grid corrosion, and oxygen recombination issues that restrict their lifespan and efficiency.
To overcome these challenges and enhance performance in modern micro-hybrid systems, detailed, real-time understanding of electrochemical processes at the electrode-electrolyte interface is required.
Raman spectroscopy has emerged as a powerful, non-destructive analytical technique capable of providing high-resolution structural and chemical information on lead-acid battery components, including lead oxides, lead sulfates, and various forms of carbon additives. Raman spectroscopy is a precise, "technique of choice" for studying the formation of lead sulfate (PbSO4) crystals, which is the primary cause of capacity loss in partial state of charge operation. Raman imaging allows for the characterization of sulfation distribution along electrode cross-sections, providing insights into uneven material degradation and inactive areas. |  |
 | Material and Method:Raman spectra were acquired using Technos IndiRAM CTR 300 Raman Spectrometer, designed to provide high spectral resolution, wavelength stability, and excellent signal-to-noise performance for Battery analysis.
Three type of battery were analysis -
These new and old battery is decided to see the effect of impurities (additive common material: BaSO4 (0.5-1%), Sb, As, Cr, Cu, and Sn)) in Raman spectrum. |
Result and Discussion:
Chemical reaction in Lead acid battery as follows:
(1) Discharging state: Product at both electrode : PbSO4
Cathode reaction: PbO2 + 3H+ + HSO4- → PbSO4 + 2H2O
Anode reaction: Pb + HSO4- → PbSO4 + H+ + 2e-
(ii) Charging state: Cathode: PbO2, Anode: Pb
Cathode reaction: PbSO4 + 2H2O → PbO2 + 3H+ + HSO4-
Anode reaction: PbSO4 + H+ + 2e- → Pb + HSO4-
Raman mode of lead sulphate (PbSO4), lead oxide (PbO2) and barium sulphate (BaSO4).
1. Raman mode of Anglesite (Pure PbSO4)
- Tetrahedra symm. bend mode: [437, 449: ref 1], [436, 439, 451: ref 2]
- Tetrahedra asymm. bend mode : [617, 640: ref 1], [605, 613, 635: ref 2 ]
- Tetrahedra symm. Stretch. of (SO4)-2: 977 [ref 1], [974, 980, 981: ref 2]
- Tetrahedra asymm. stretching mode: [1051, 1140, 1157: ref 1], [1057,1060, 1065 : ref 2], [1148, 1154, 1160: ref 2] cm-1
2. Raman mode of lead oxides
- PbO2: eg : 424 cm-1, a1g: 515 cm-1 and b2g :653 cm-1 [ref 3 ]
- PbO: 117, 139, 270, 314, 500, 533
- Pb3O4: 122, 148, 540-550
3. Raman mode of Barite (Pure BaSO4 : ref 4)
- Symmetry bend vibration at ~ 450 cm–1
- Asymmetry bend vibration ν4(F2) at ~ 620 cm–1,
- Symmetric stretching of [SO4]-2 at ~ 988-1000 cm-1
Asymmetric stretching at ~1080, 1150, 1167 cm–1
Peak Assignment:
Scooter old /dead battery:
Conclusion: Mainly PbSO4 with small amount of BaSO4, PbO, PbO2, Pb3O4, Cr2O3, Cr8O21, Fe2O3, Fe3O4.
UPS old/dead battery :
Conclusion: Mainly BaSO4 with PbSO4
Portable electronic new battery:
Conclusion: Mainly PbSO4 with small amount of PbO, PbO2 | 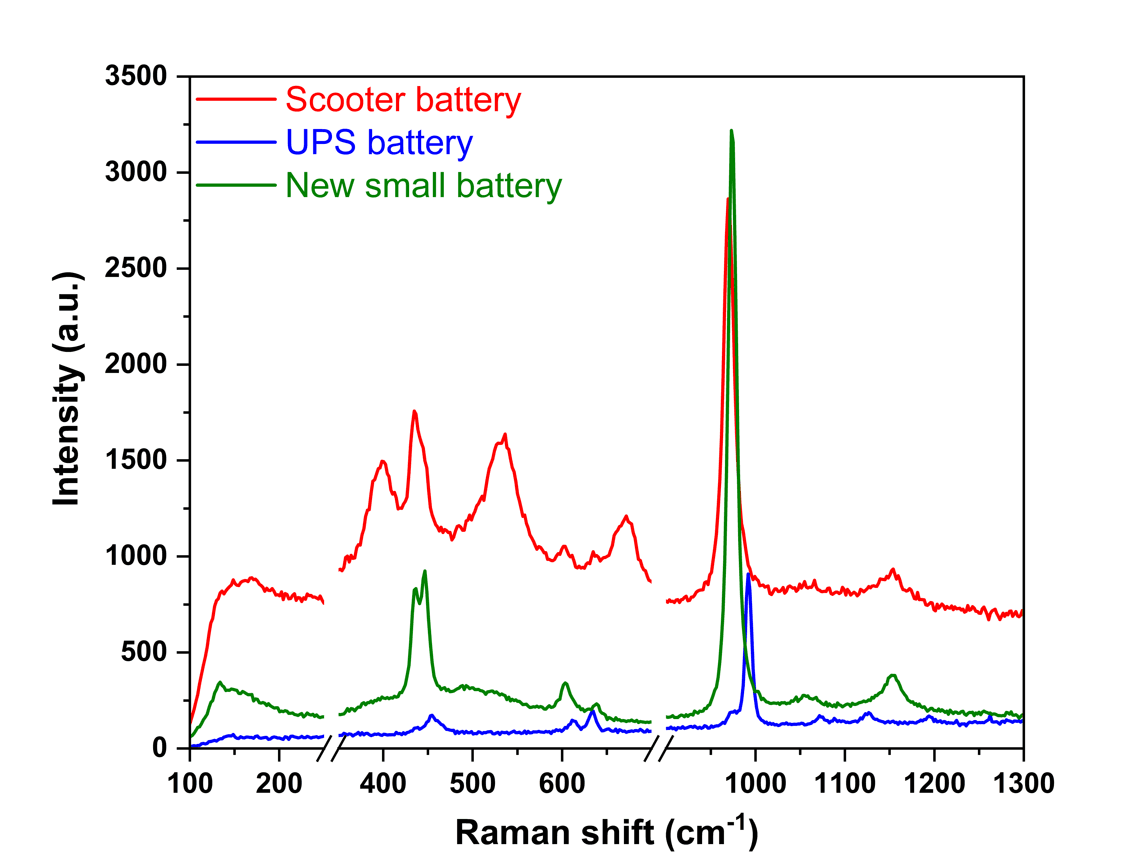 |
Conclusion:
Raman spectroscopy analysis revealed that the scooter battery sample was primarily composed of PbSO₄, along with minor amounts of BaSO₄, various lead oxides (PbO, PbO₂, Pb₃O₄), chromium oxides (Cr₂O₃, Cr₈O₂₁) and iron oxides (Fe₂O₃, and Fe₃O₄). The UPS battery sample mainly contained BaSO₄ and PbSO₄, whereas the small portable electronic battery sample was predominantly composed of PbSO₄ with trace amount of lead oxide (PbO and PbO₂). Raman spectroscopy provides a fast, non-destructive, and highly specific method for identifying Lead acid battey. The distinct spectral signatures of impurity of BaSO4 and other products demonstrate Raman’s effectiveness for Lead acid battery in exploration and energy application.
Using high-resolution IndiRAM CTR 300 Raman Spectrometer and with ongoing development of Portable Raman solutions, TechnoS Instruments enables accurate, Lead acid battery identification to support the growing demand for critical % Barium sulphate.
References
1. Krista Sawchuk, Earl F. O’Bannon III, Cara Vennari, Abby Kavner, Elise Knittle, Quentin Williams, Physics and Chemistry of Minerals (2019) 46:623–637.
2. G.L.J. Trettenhahn, G.E. Nauer and A. Neckel, Vibrational Spectroscopy (1993), 5 85-100.
3. Lucia Burgio, Robin J. H. Clark and Steven Firth, Analyst (2001), 126, 222–227
4. V. E. Shukshina, P. P. Fedorova, and M. E. Generalov, Russian Journal of Inorganic Chemistry (2019), 64,1442–1445.